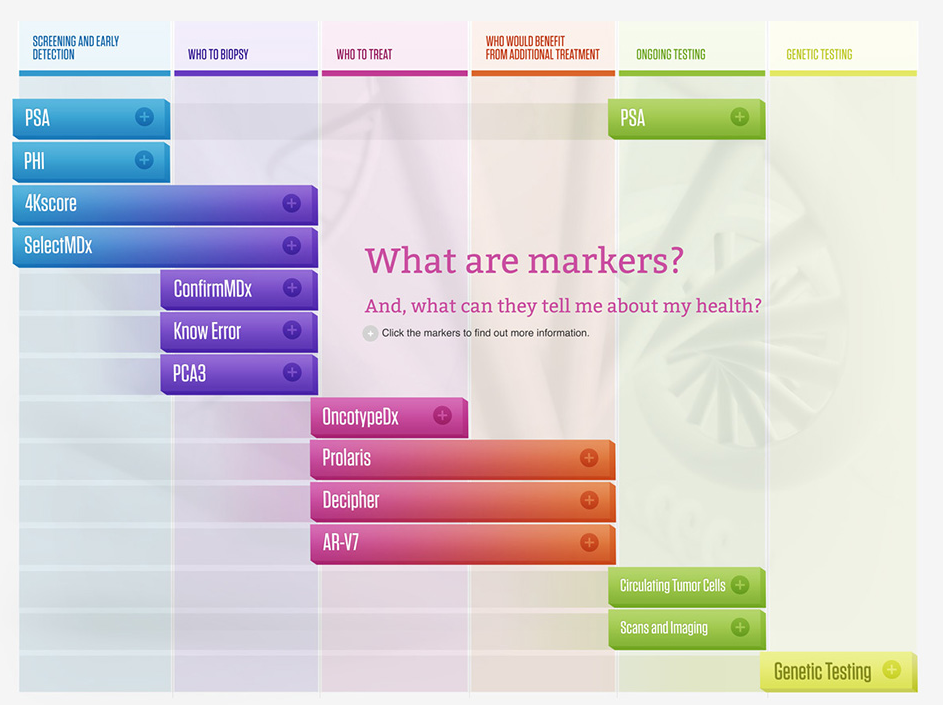
Prostate cancer markers are making an impact on the world of prostate cancer by helping to individualize patient care in the early detection, diagnosis and treatment choices for prostate cancer. Through advanced science researchers from many different institutions and companies have developed a variety of tests that look at each persons genes or biomarkers and help to determine individual risk for having prostate cancer, the need for biopsies or repeat biopsies and often the best course of treatment.
PCEC Chairman Dr. David Crawford discusses the role of Biomarkers for Primary Care Physicians. Click here to see his article!
Click Here for the Prostate Marker Map
Markers & Tests for Early Detection of Prostate Cancer
-
PSA: Prostate-specific antigen, or PSA, is a protein produced by cells of the prostate gland. The PSA test measures the level of PSA in a man’s blood. PSA is present in small quantities in the serum of men with healthy prostates, but is often elevated in the presence of prostate cancer or other prostate disorders.
- SelectMDx: Helps identify patients at increased risk for aggressive disease, thereby aiding in the selection of men for prostate biopsy.
- phi: The Prostate Health Index (phi) is a new combination of 3 blood tests that results in a score, or “phi score.” This score gives you more accurate information on what an elevated PSA level might mean and the probability of finding cancer on biopsy.
- 4Kscore: The 4Kscore Test is a blood test that provides a patient-specific probability for finding a aggressive (Gleason score 7 or higher) prostate cancer upon biopsy. The information can be used by the Urologist to have an informed discussion with the patient about whether or not to have a prostate biopsy. The 4Kscore Test measures four prostate-specific kallikreins in the blood: Total PSA, Free PSA, Intact PSA, and Human Kallikrein 2 (hK2). The blood test results are combined in an algorithm with patient age, digital rectal exam (nodule, no nodule), and prior negative biopsy (yes, no). The 4Kscore Test then provides a % probability on a scales from <1% to >95% for the patient having aggressive prostate cancer.
- ProgensaPCA3: A FDA-approved urine test that detects the over-expression of a the PCA3 gene, which is specific to prostate cancer and an accurate predictor of whether cancer may be present. The PCA3 score is used to determine if a repeat biopsy is needed in men who are used to determine a man’s PCA3 score, which indicates the need for biopsy. Research is underway looking at PCA3 as prostate cancer screening population as well.
- ERG Protein Tissue Marker: Development of an ERG protein assays to be utilized on prostate cancer biopsy tissue is also underway. Presence of the ERG protein in tissue helps to identify patients who have prostate cancer. Additionally, the presence of ERG in high grade PIN (pre-cancerous lesion) is indicative of a patient more likely to be diagnosed with cancer upon the next biopsy.
Markers & Tests for Determining Who to Repeat Biopsy
- ConfirmMDx: For men with an initial negative biopsy, independently published clinical studies have shown that the ConfirmMDx test is the most significant, independent predictor of patient outcome relative to other available clinical factors such as age, PSA and DRE results.
- PCA3: If you are concerned about prostate cancer because of an elevated PSA or are feeling insecure about a previously performed (negative) biopsy, the PCA3 urine test can provide additional information that may help you and your physician to decide whether a (new) biopsy is really needed.
- 4Kscore: The 4Kscore Test is a blood test that provides a patient-specific probability for finding a aggressive (Gleason score 7 or higher) prostate cancer upon biopsy. The information can be used by the Urologist to have an informed discussion with the patient about whether or not to have a prostate biopsy. The 4Kscore Test measures four prostate-specific kallikreins in the blood: Total PSA, Free PSA, Intact PSA, and Human Kallikrein 2 (hK2). The blood test results are combined in an algorithm with patient age, digital rectal exam (nodule, no nodule), and prior negative biopsy (yes, no). The 4Kscore Test then provides a % probability on a scales from <1% to >95% for the patient having aggressive prostate cancer.
Markers & Tests for Determining Who to Treat for Prostate Cancer
- Oncotype DX: The Oncotype DX Prostate Cancer Assay harnesses the power of genomics to provide a more precise and accurate assessment of risk based on individual tumor biology.
- Prolaris: Prolaris is a measure of how fast a prostate cancer tumor is growing. Biopsy tissue samples can be used to determine a patient’s personal Prolaris Score. Studies have shown that Prolaris provides an accurate assessment of cancer aggressiveness.
- P10: "Most studies have found that PTEN loss is a powerful predictor of which prostate tumors are likely to recur or metastasize," says urologic pathologist Tamara Lotan, M.D.
- ProMark: On September 10, 2013, Metamark announced positive results from a large clinical validation study of ProMarkTM, its biopsy-based prostate cancer prognostic test. The study met its primary endpoint demonstrating that ProMarkTM, evaluated in standard formalin-fixed, paraffin-embedded prostate tissue, can differentiate indolent from aggressive disease.
Markers & Tests for Determining Who Would Benefit from Additional Treatment
- Prolaris: Prolaris is a measure of how fast a prostate cancer tumor is growing. Biopsy tissue samples can be used to determine a patient’s personal Prolaris Score. Studies have shown that Prolaris provides an accurate assessment of cancer aggressiveness.
- Decipher: The Decipher test predicts the probability of metastasis after surgery and provides an independent assessment of tumor aggressiveness; information distinct from that provided by Gleason score or PSA.
Markers & Tests for Ongoing Testing
- PSA: Prostate-specific antigen, or PSA, is a protein produced by cells of the prostate gland. The PSA test measures the level of PSA in a man’s blood. PSA is present in small quantities in the serum of men with healthy prostates, but is often elevated in the presence of prostate cancer or other prostate disorders.
- Testosterone: In men, testosterone plays a key role in the development of male reproductive tissues such as the testis and prostate as well as promoting secondary sexual characteristics such as increased muscle, bone mass, and the growth of body hair.
- Circulating Tumor Cells: Circulating tumor cells (CTCs) are cells that have shed into the vasculature from a primary tumor and circulate in the bloodstream. CTCs thus constitute seeds for subsequent growth of additional tumors (metastasis) in vital distant organs, triggering a mechanism that is responsible for the vast majority of cancer-related deaths.
Markers & Tests for Genetic Testing
- Genetic Testing: Genetic counseling before and after genetic testing is very important. Genetic testing plays an important role in identifying who is at risk for certain disease, who is at risk for more aggressive disease and often genetic testing can play a critical role in determining the best and most effective treatment options. In prostate cancer specifically, identifying genetic mutations of inherited prostate cancer has implications for a man's risk of prostate cancer, the risk of more aggressive disease and to personalize treatment options. Additionally, there may be an impact on families, if certain gene mutations are found it may be important that family members are also tested.
To learn more about Prostate Cancer Markers, visit www.prostatemarkers.org